The Spike protein and its impact in the cholinergic system
The Intricacies of Spike Protein Interactions
In the process of writing this substack, I accidentally found a remarkable paper, I will address these changes and remediate/ameliorate the changes, which are more pertinent in Long Covid and neurological conditions.
Back in 2020, after much observation, research, and even talking to clinician friends and brainstorming I advised most people to take choline supplements or eat eggs. At some point in early 2021, I found out, incidentally, the Chinese government demanded doctors prescribe a standard treatment to anyone with a positive test for Covid, and among the prescribed items there were “4 eggs per day”.
I will make clear why and how in a short bit.
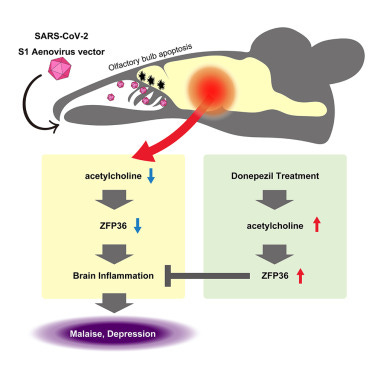
SARS-CoV-2 S1 protein causes brain inflammation by reducing intracerebral acetylcholine production
Highlights
Mice expressing S1 protein in the nasal cavity showed decreased acetylcholine production.
In the brain, ZFP36 was decreased and inflammatory cytokine expression was enhanced.
The mice exhibited malaise and depressive clinical signs.
Donepezil normalized enhanced inflammatory cytokine production and mitigated depression.
At the start of the paper, the authors raise a point I did for months (to fall on deaf ears, for the most part, narrative pushers ignore evidence) autopsies from patients who died from Covid-19 had tissue degeneration and damage in the brain but lacked evidence of central nervous system damage directly from SARS-CoV-2 pointing out that the brain-related symptoms such as cognitive impairment must have other mechanisms. Another point raised by the authors is olfactory system cell damage, in particular, olfactory bulbs resulting in increased intracellular calcium in olfactory epithelial cells. If you missed my early mitochondria-related substacks, intracellular calcium levels are extremely important for cell function, and fluctuation often leads to problems., they cite their own previous research which demonstrated Human Herpesvirus 6 (HHV-6) protein SITH-1 doing something similar.
It is rather interesting because, alongside Epstein Barr, CMV, HHV-6 is another one of the pathogens commonly responsible for a decent portion of Long Covid, especially the ones with neurological symptoms.
Intracellular calcium elevating activity of S1 protein
When spike protein invades cells, it is cleaved into S1 and S2 subunits. Using mouse cells, we found that the N-terminal domain (NTD) was responsible for calcium-elevating activity on S1 protein . S1 protein also elevated intracellular calcium in human cells (A549)
We once again find another mechanism by which the same section of the virus induces a pathological effect, in this case, the NTD. The authors do not separate the effects of the NTD from the S1 most likely because the NTD is part of the S1, but I personally will remind the reader a few times here, to keep this in mind. To rest the effect of the S1 in elevating intracellular calcium on olfactory bulbs in vivo (live animals) the authors created an adenovirus vectored S1 that doesn’t replicate, using the original Wuhan Strain, and inoculate mice with it, one week after they tested the behavior and the genes in these mice.
S1 expressing mouse manifests olfactory bulb damage, malaise, depression and brain inflammation
In addition, using histologic samples of the whole brain without the olfactory bulb, we observed enhanced expression of inflammatory cytokines (IL-6, TNFα) and a chemokine (CCL-2) with an inflammation-promoting function suggesting that inflammation had occurred in the brain.
We also observed enhanced expression of IL-6 in the lungs. In the lungs, a correlation of inflammatory cytokine production with S1 mRNA expression ) suggested that inflammation in the lungs was due to the inflammation-inducing action of S1 protein In contrast, since S1 mRNA expression was not detected in the brain, this suggested that inflammation in the brain was due to an indirect effect
Besides testing the mice's behavior and finding clinical signs of depression in the model they used, the histological findings are more in line with my preferred method of measurement. By using the whole brain, without the olfactory bulb, they observed the expression of the usual suspects in both neuroinflammatory and neurodegenerative conditions, IL-6 and TNF-Alpha, beside CCL-2 (Chemokine), the 3 being inflammation-promoting proteins. Also observed was IL-6 in the lungs which correlated with S1 mRNA expression, suggesting in this study that the lung inflammation was due to the inflammation-inducing property of the S1, no S1 mRNA was found in the brain.
Since the results of their tests suggested an association between S1 (the NTD) to the increase in calcium signaling activity and pathogenicity they measured this effect between Wuhan, Alpha, and Delta Strain, and as expected, Delta had the strongest effect, followed by Alpha and Wuhan.
While outside the scope of the author’s observations, I must remind you that there is now solid evidence of the presence of Spike (and S1) in the brain. What they describe here and following is but another mechanism for injury mediated by the Spike.
Impairment of acetylcholine’s anti-inflammatory effect was associated with brain inflammation in S1 mouse
To understand the exact pathway that olfactory bulb damage from S1 (NTD) leads to damage to the brain and body (lungs in this papers), they focused on neurotransmitters in neurons connecting to the olfactory bulb. Finding a decrease in acetyltransferase (ChAT) an enzyme responsible for synthesizing (creating) acetylcholine in the brain, so no surprise that the levels of acetylcholine were found to be decreased as well.
Since acetylcholine participates in a very important anti-inflammatory response called Cholinergic Anti-inflammatory Pathway (CAP), a response to suppress inflammation in peripheral tissues and in the brain. By using a α7nAchR chemical agonist (inducing the body to produce more of) they found this normalized the excessive inflammatory response produced by S1. α7nAchR is one of the most important receptors of the nACh family and is essential in the anti-inflammatory function of the CAP, it also plays a significant role in the immune system. The normalization after this agonism indicates that brain inflammation in the S1 mouse is a direct byproduct of CAP disruption.
Following this they uncovered how S1 lowers the expression of ZFP36 (Zinc Finger Protein), a protein responsible to bind to mRNA so the body degrades (dissolves, eats away) that mRNA, it is an inflammatory controlling protein. Increasing α7nAchR enhanced ZFP36 and lowered TNF Alpha, thus lowering inflammation and combating the S1 inflammatory properties. Here is where things get a little complicated from my perspective.
Acetylcholine esterase inhibitor donepezil improves brain symptoms
The acetylcholine esterase inhibitor donepezil is a central cholinergic agent used clinically for the treatment of dementia. To investigate the possibility of its repurposing for the treatment of neurological complications in COVID-19, we investigated symptom improvement due to donepezil in the S1 mouse.
Administration of donepezil resulted in normalizing inflammatory cytokines (IL-6, TNFα) that had been enhanced by S1 protein). Since IL-6 and TNFα genes are targets of ZFP36, this finding suggests that donepezil brought about a recovery in CAP. However, administration of donepezil did not mitigate the enhanced inflammatory cytokine production in the lungs
In simple terms, Donepezil stops your body from breaking down Acetylcholine (ACh), thus more ACh is available to bind to α7nAChR and exert its anti-inflammatory effects, and given the properties of Alpha 7 and ACh itself, it enhances the activation of the CAP and positively modulates immune responses. This can result in dampened inflammation and reduced immune-mediated tissue damage.
The authors suggest that Donepezil should be considered as a treatment option for neurological complications after SARS-CoV-2 infection. The substack below is both highly informative and pertinent to the subject we just discussed.
And to be abundantly clear, no I do not think everyone should just start using nicotine (either gum or patches) but it is something to have in mind and in your toolbox in very specific circumstances, quite a few people do benefit from the use, especially the ones with specific neurological conditions, or specific (although complex) chronic infections/inflammation, among these Long Covid. There are other ways which I will mention after the next, significant paper.
To remind you why I somewhat focused and kept mentioning the NTD, the N-Terminal Domain of SARS-CoV-2 may also be referred to by another name. A Galectin-fold. Or a Galectin-3 mimic. This is significant because Galectin-9 will directly interact with some AChRs, and 3 will do much more indirectly. Add this to the growing list of interactions.
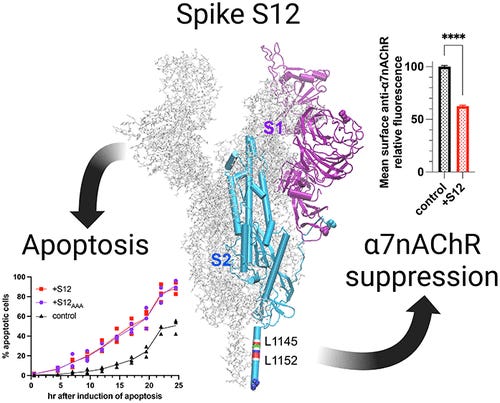
SARS-CoV-2 Spike Protein Downregulates Cell Surface α7nAChR through a Helical Motif in the Spike Neck
Note - Spike S12 reads as Spike S One Two, not twelve =P
S12 can act alone or in conjunction with other mediators on target cells, stimulate different cell types, damage the integrity of the blood–brain barrier, and contribute to the pathogenesis of long COVID. (10,12−15) These findings undoubtedly implicate S12 as an inducer of cellular dysfunction.
The α7 nicotinic acetylcholine receptor (α7nAChR) was linked to COVID-19 pathophysiology at the early stage of the pandemic. (16) α7nAChR forms homopentameric ligand gated ion channels that mediate synaptic transmission in the central and peripheral nervous systems. (17,18) It is involved in cognitive function, mental health, and neurodegenerative diseases. (19) α7nAChR is also a major player in the cholinergic anti-inflammatory pathway, (20,21) which attenuates proinflammatory cytokine production and minimizes tissue and organ injury during inflammation. The α7nAChR agonists, such as nicotine, are essential for initiating the cholinergic anti-inflammatory pathway and effective in reducing macrophage cytokine production and inflammation. (22,23) Therapeutic implications of cholinergic signaling in acute and chronic pathology, including a therapeutic avenue for treating COVID-19, have been supported by human data and animal studies. (24−26) α7nAChR is widely expressed across the human body in both neuronal and non-neuronal cells. (19,24,27−30) In the brain, it is expressed on both pre- and postsynaptic membranes and particularly in regions implicated in cognitive function, such as the hippocampus and cortex. (31,32) α7nAChR is also expressed in immune cells, such as macrophages, that form the basis for some of the known α7nAChR-mediated anti-inflammatory effects. (27) A deficiency of functional α7nAChR is implicated in neuropsychic diseases and disrupts the cholinergic anti-inflammatory pathway. (30,33−38)
The study uncovers significant discoveries about the interplay between the SARS-CoV-2 Spike protein and the α7 nicotinic acetylcholine receptor (α7nAChR), bearing much significance to what we just discussed in a causative form on the physiological and immunological functions discussed above.
One crucial finding is the profound downregulation of surface α7nAChR by the spike protein's S12 region, this being an unexpected finding since the proposed mechanism beforehand was via the Furin Cleavage Site (FCS) and potential sites mimicking toxins (which I don’t agree with to this day). The extent of this downregulation is remarkable, affecting more than one-third of surface α7nAChR and over half of the functional α7nAChR. Importantly, this downregulation occurs in different cell types, as observed in both neuronal PC12 cells and HEK293 cells expressing native and recombinant α7nAChR, respectively. Given that α7nAChR is expressed in various cells the downregulation induced by S12 is likely widespread across different cell types.
The depletion of functional α7nAChR caused by the S12 region can have negative consequences on neuronal circuitry, anti-inflammatory signaling, and immune response. Deficiencies in functional α7nAChR in the hippocampus and other brain regions have been associated with neuropsychiatric disorders characterized by cognitive impairments. On the other hand, enhancing α7nAChR function has been shown to improve cognitive performance, attention, and memory. Furthermore, α7nAChR plays a vital role in the cholinergic anti-inflammatory pathway, and its presence in immune cells is critical for α7nAChR-mediated anti-inflammatory signaling and neuroprotection. A significant reduction in α7nAChR will not only impact cognitive function but also impair normal immune responses and much more, to be discussed below. These findings provide a new mechanistic perspective in understanding the cognitive and immune dysfunction symptoms observed in Long Covid individuals
Another significant discovery is the underlying mechanism by which the S12 region downregulates surface α7nAChR. The helical segment (L1145-L1152, in the image above) ) in the spike protein's neck region shares homology with the α7nAChR helical segment (L411-V418) responsible for binding chaperones that aid in receptor assembly and trafficking (building and moving). By analyzing the binding interactions, it was observed that S12 competes with α7nAChR chaperones (RIC-3 and antiapoptotic Bcl-2 proteins), leading to the downregulation of surface α7nAChR. This competition mechanism explains why the coexpression of S12 does not affect the surface population of another receptor type, α4β2nAChR, which lacks certain chaperone-binding sites, but affects α7nAChR.
The conservation of the L1145-L1152 segment in all SARS-CoV-2 variants is noteworthy, this is a part of the virus that doesn’t particularly change among all variants. All Spike-centric vaccines, including mRNA and Adenovirus ones all possess this section, they all have the capacity to engage in this mechanism and the authors suggest mutating this segment in the next generation of vaccines employing the Spike to avoid these effects, something they did in this paper to test their hypothesis. I will leave you with the author’s closing remarks before delving deeper into much of what the α7nAChR function entails.
α7nAChR is a very important receptor, its significance in physiological functions goes beyond neuroprotection, dampening of inflammatory signals and inflammation itself, and the immune system. Its activation is protective in aneurysms by inhibiting the same pathway SARS-CoV-2 often engages, therefore its substantial lack of presence in the cell surface negatively impacts the aforementioned mechanism. Activation of α7nAChR reduces the Th17 responses in CD4 cells, the very mechanism by which a lot of the autoimmune damage and disease, and pathogen-mediate autoimmunity are created, the same mechanism that is the dominant response in the mRNA vaccines, in fact, this may be an argument for the Th17 response in the SARS-CoV-2 vaccine.
Its activation alleviates psoriasis-like inflammation by inhibition of STAT3 and NF-kB signaling pathways, a powerful inflammatory pathway in the body, one responsible for a lot of the subsequent damage from a severe infection. Via a different pathway but using STAT3 and HO-1, the activation of Alpha 7 preserves intestinal barrier integrity, integrity that is compromised in many infected people, from mild to severe infection, and a common denominator among a significant portion of Long Covid.
It regulates the release of TNF-Alpha in microglia (the immune cells of the brain) via a G-coupled protein, a topic I am yet to cover and write about, but significant in regards to Long Covid and autoimmunity from SARS-CoV-2 in many levels. Its significance here is neuroprotection.
It also plays a role in the release of Nitric Oxide, and modulating blood pressure, NO deficiency, or improper release of is something seen in many of the Long Covid, and cardiovascular injured.
α7nAChR also plays a role in both the circulatory and capillary (microcirculatory in simplistic terms) systems. And for last, I couldn’t forget that α7nAChR directly influences the activities of enzymes involved in the Kynurenine Pathway, both of these pathways acting upon each other to keep a balance between protection and toxicity (toxicity in regards to kynurenine metabolites especially), abnormalities on the KP and dysfunction of α7nAChR contributes to neuroinflammatory, neurodegenerative diseases, and neurotransmitter imbalances observed in many neurocognitive pathologies.
Further reading on the significance of the dysregulation of the Kynurenine Pathway.
Now besides Nicotine, which isn’t exactly a very “marketable” choice, there are a few things may do or consume in regards to α7nAChR and its Cholinergic pathway.
eating eggs, eggs are a superfood, and long-term they are beneficial, especially allied with a good diet not rich in carbohydrates and processed foods
third, supplementing choline, which has a myriad of positive effects choline bitartrate or alpha-GPC (Alpha-Glycerophosphocholine) being the most common, other choices are CDP-Choline (Citicoline), Bacopa monnieri, and Phosphatidylcholine (this is already found in eggs)
As a last note, given I recently wrote about biofilms, here is an interesting tidbit of information. Acetyl Choline can directly interact with and inhibit Candida Albicans biofilm formation and also promote an efficient immune response against the pathogen, and plays a significant role in sepsis.
This may be a topic I may go back to, not solely in regards to SARS-CoV-2, but in general.
I wish you all a happy weekend, and for anyone who chose to support my work, thank you !